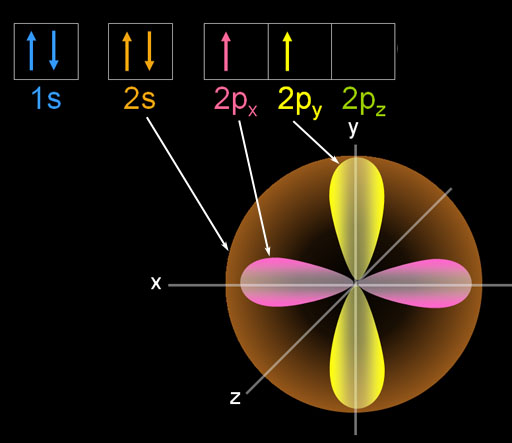
This results in beautiful geometric structures called orbitals that represent the distinct regions around the nucleus that each electron traces out. While these electrons all stick within the atom because of their attraction to the protons, they also mutually repel each other, causing them to spread out around the nucleus in regular patterns. As we know, the positively-charged protons in the nucleus of an atom tend to attract negatively-charged electrons. It also describes how these concepts apply to the work that the Department of Energy’s Office of Science conducts as it helps the United States excel in research across the scientific spectrum.Electron configurations are a simple way of writing down the locations of all of the electrons in an atom. DOE Explains offers straightforward explanations of key words and concepts in fundamental science.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
